
What are allergies?
Allergies, also called hypersensitivity reactions, are responses of the immune system (the defenses of our body) to come into contact with substances called allergens which are usually tolerated by most people. These are frequent pathologies that affect more than 25% of the world’s population according to the World Health Organization (WHO). In addition, over the last several decades, changes in the environment, habitat and way of life and a greater number of pets have favored the progression of allergy. Immunotherapy, also known as hyposensitizing therapy, reduces the sensitization of patients by attenuating the symptoms they experience in contact with the allergen. Immunotherapy is usually effective, the improvement is observed a few months after having started the vaccination, becoming apparent around the first year of treatment.
Allergy treatment
Treatment with allergen vaccines consists of progressively applying repeated doses of the allergen to which one is allergic in order to diminish or eliminate the reaction that occurs when coming into contact with it. Allergenic vaccines are able to modulate the patient’s immunological response, thereby changing the natural course of the allergic disease mediated by the production of IgE antibodies, with a long-term lasting effect after the end of treatment. This type of immunotherapy is used to treat allergy cases caused by pollen, dust mites, animal epithelia and certain fungi.
Apither SCIT
Efficacy and subcutaneous safety
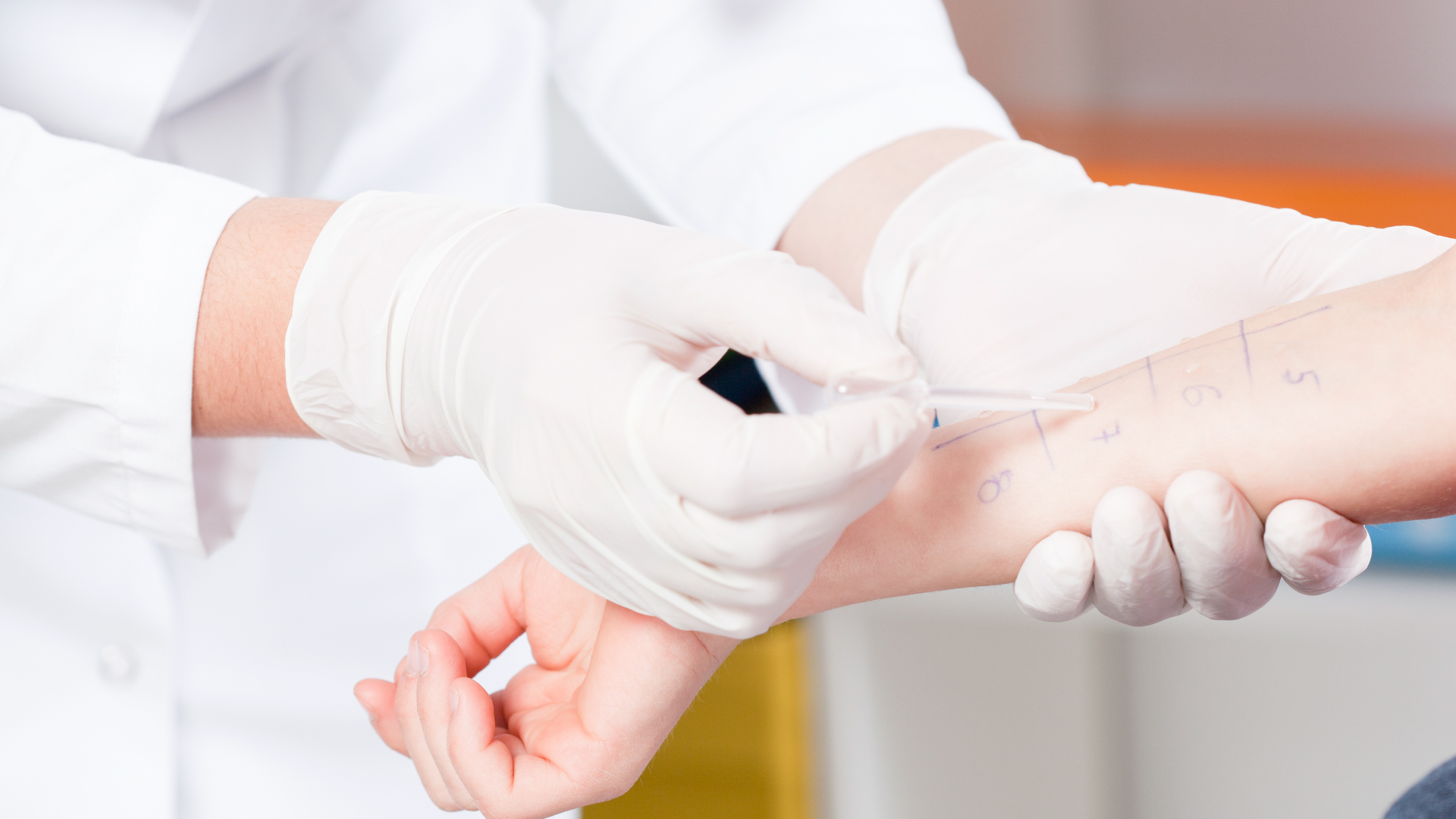
The subcutaneous route of administration consists of periodic injections of allergens at the subcutaneous level, usually on the inside of the arm, above the elbow but never intravenously or intramuscularly.
The subcutaneous treatment consists of applying a weekly starter treatment and a monthly maintenance treatment. The duration of the starter and maintenance will depend on the type of subcutaneous treatment selected and the frequency set by the specialist.
Subcutaneous vaccines are prepared individually for each patient because each person has a different sensitization to certain allergens, so it is up to the doctor to assess the composition of the SCIT vaccine in each case.
Depending on the chemical composition, we differentiate between POLYMERIZED and DEPOT:
Apither Depot
Apither DEPOT is an immunotherapy treatment (vaccine) administered subcutaneously and made up of allergenic extracts adsorbed in aluminum hydroxide, thus facilitating the slow and gradual absorption of the antigen by the organism to which the patient is sensitized.
Apither DEPOT is prepared individually for each patient, because each person has a different sensitization to certain allergens, so it is up to the doctor to assess the composition of APITHER Depot on a case-by-case basis.
The benefits of Apither DEPOT are:
Apither Depot is a subcutaneous vaccine used for the treatment of allergic disease that occurs with rhinitis, rhinoconjunctivitis or seasonal or perennial bronchial asthma.
Starter treatment:
• Vial 1:
Maintenance treatment:
• Vial 3:
The following formats are available:
· Starter formats:
· Maintenance formats:
Dermatophagoides pteronyssinus
Dermatophagoides farinae
Dermatophagoides MIX (D. pteronyssinus, D. farinae)
Blomia tropicalis
Lepidoglyphus destructor
GRAMINEAS:
Wild-spontaneous Gramineas (Dactylis, Lolium, Phleum, Poa)
Cultivated grasses (Hordeum, Secale, Triticum)
Dactylis glomerata
Lolium perenne
Phleum pratense
Poa pratensis
Hordeum vulgare
Secale cereale
Cynodon dactylon
TREES:
Olea europaea
Cupressus arizonica
Platanus acerifolia
Fraxinus excelsior
PLANTS:
Artemisia vulgaris
Chenopodim album
Jewish Parietaria
Salsola kali
Plantago lanceolata
EPITHELIUM:
Cat
Dog
FUNGI:
Alternaria alternata
Aspergillus fumigatus
Penicillium notatum
Polymerized Apither
Polymerized Apither consists of a sterile suspension composed of allergens to which the patient is sensitized, extracted fractionally, purified by ultrafiltration, evaluated by immunochemical techniques, polymerized with glutaraldehyde and diluted in physiological saline for specific immunotherapy.
This vaccine is formulated with polymerized allergens. Chemically modified allergens show a reduced allergenicity, so they show better tolerance, thus lowering the likelihood of adverse reactions.
The benefits of POLYMERIZED Apither are:
APITHER POLYMERIZED is a subcutaneously administered vaccine indicated for hyposensitizing immunotherapy in allergies with allergic rhinitis, allergic rhinoconjunctivitis or seasonal or perennial bronchial asthma.
Starter treatment::
Vial 1: · Labeling
Vial 2: · Labeling
Maintenance bottle:
Vial 1: ·Labeling
The following formats are available:
Starter formats:
Maintenance formats:
Dermatophagoides pteronyssinus
Dermatophagoides farinae
Blomia tropicalis
Lepidoglyphus destructor
GRAMINEAS:
Wild-spontaneous Gramineas (Dactylis, Lolium, Phleum, Poa)
Perennial lolium
Cynodon dactylon
TREES:
Olea europaea
Cupressus arizonica
Platanus acerifolia
PLANTS:
Artemisia vulgaris
Chenopodim album
Jewish Parietaria
Salsola kali
Plantago lanceolata


